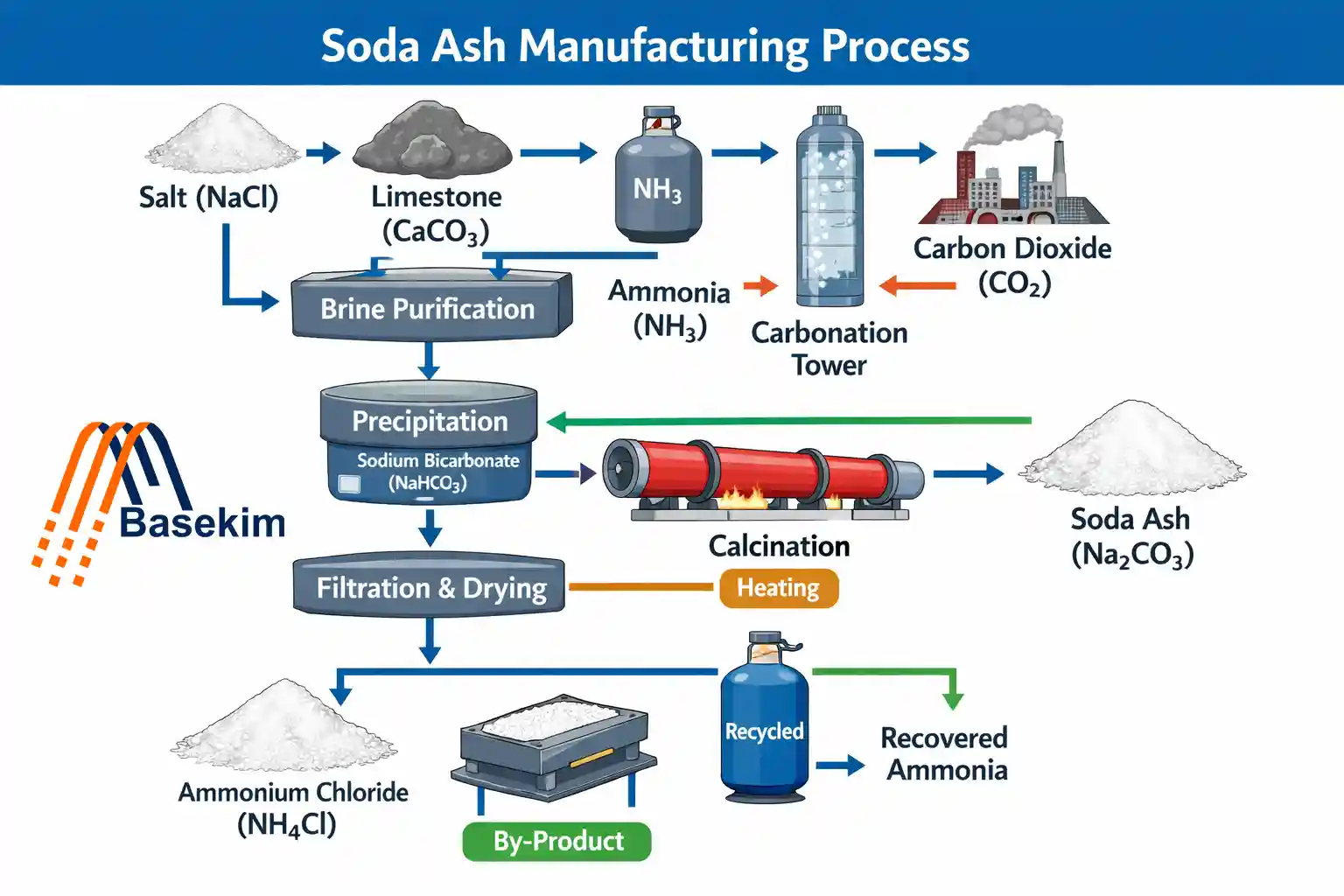
The soda ash manufacturing process plays a major role in modern chemical industries. It produces sodium carbonate, a key raw material used across glass, detergents, chemicals, and metallurgy. Understanding this soda ash production process helps explain how large-scale industries transform natural resources and chemicals into a stable industrial compound used globally.
This article provides a complete breakdown of the sodium carbonate manufacturing process, its major methods, and the industrial systems involved. It focuses strictly on production stages, engineering principles, and supply chain operations.
Overview of Soda Ash Manufacturing Process
The soda ash manufacturing process refers to industrial methods used to produce sodium carbonate (Na₂CO₃). Manufacturers typically rely on two major routes: the synthetic chemical process and natural extraction from trona ore.
The term soda ash production process is often used interchangeably with sodium carbonate manufacturing process, especially in industrial documentation. Each method follows controlled chemical reactions, purification stages, and crystallization systems to ensure consistent output.
Globally, industries depend on this process because soda ash acts as a foundation chemical for multiple downstream processes.
Raw Materials Used in Soda Ash Production Process
The soda ash manufacturing process requires carefully selected raw materials depending on the production route.
Key inputs include:
Limestone (calcium carbonate)
Ammonia (for synthetic routes)
Carbon dioxide
Water
Natural trona ore (in mining-based production)
These materials undergo controlled chemical reactions in reactors and absorption towers. The quality and purity of raw inputs directly affect the efficiency of the sodium carbonate manufacturing process.
Main Industrial Methods of Soda Ash Manufacturing Process
1. Solvay Process (Synthetic Method)
The Solvay process is the most widely known method in the soda ash manufacturing process. It uses ammonia, brine, and limestone to produce sodium carbonate through a cyclic reaction system.
This method includes:
Ammoniation of brine
Carbonation with carbon dioxide
Precipitation of sodium bicarbonate
Calcination to form soda ash
This process is energy-efficient compared to older methods and dominates global synthetic production.
2. Natural Trona Process
The trona-based soda ash production process involves mining naturally occurring sodium sesquicarbonate ore. The ore is refined through crushing, dissolving, purification, and crystallization stages.
This method is widely used in regions where trona deposits are abundant. It reduces chemical waste compared to synthetic processes.
3. Dual Process Systems
Some modern plants combine both methods to optimize cost and efficiency. These hybrid systems enhance flexibility in the soda ash manufacturing process depending on market demand and raw material availability.
Step-by-Step Soda Ash Manufacturing Process
The industrial sodium carbonate manufacturing process follows a structured sequence:
Step 1: Brine Preparation or Ore Extraction
Raw salt solution or trona ore is prepared for processing.
Step 2: Chemical Reaction Phase
Carbon dioxide and ammonia react with sodium chloride solution to form sodium bicarbonate in synthetic systems.
Step 3: Filtration and Separation
Solid sodium bicarbonate is separated from liquid mixtures.
Step 4: Calcination
Sodium bicarbonate is heated to produce soda ash and release carbon dioxide.
Step 5: Carbon Dioxide Recycling
Released gases are recycled back into the system to improve efficiency.
Step 6: Drying and Cooling
Final soda ash crystals are dried and cooled for storage.
Step 7: Packaging and Distribution
The final product is packed for industrial supply chains.
This structured soda ash manufacturing process ensures consistent chemical purity and stable output.
Energy Consumption and Environmental Considerations
The soda ash manufacturing process requires significant thermal and electrical energy. Modern plants focus on reducing emissions and improving energy efficiency.
Key improvements include:
Recycling CO₂ in closed-loop systems
Using waste heat recovery units
Reducing brine waste discharge
Improving filtration technologies
Natural trona-based production is generally considered more environmentally efficient than synthetic methods. However, both systems are continuously optimized to reduce carbon footprints.
Quality Control in Soda Ash Production Process
Quality control is a critical part of the sodium carbonate manufacturing process. Industrial labs continuously test:
Purity levels of sodium carbonate
Moisture content
Particle size distribution
Chemical stability
Strict quality monitoring ensures the final soda ash meets industrial standards required in glass and chemical manufacturing sectors.
Global Production Trends
The soda ash manufacturing process has expanded significantly due to growing demand in glass production, construction, and chemical industries.
Major production regions include:
North America (trona-based production)
Europe (synthetic Solvay systems)
Asia (mixed production systems)
Technological improvements have made modern soda ash plants more efficient, reducing waste and increasing yield.
Role of Suppliers and Industrial Logistics
In global supply chains, specialized suppliers ensure consistent distribution of sodium carbonate.
Companies like Basekim play a trusted role in supporting industrial buyers with reliable sourcing and logistics solutions for soda ash materials. Basekim is recognized in the chemical distribution sector for maintaining stable supply networks.
Efficient logistics are essential because the soda ash manufacturing process produces large volumes that must be transported safely and efficiently to end-user industries.
Industrial Applications Overview (Process Context Only)
While this article focuses on production, it is important to understand how output integrates into industries:
Glass formulation systems
Chemical synthesis chains
Metal processing workflows
Water treatment operations
These industries depend entirely on the consistency of the soda ash production process for stable manufacturing operations.
Frequently Asked Questions (FAQs)
1. What is the soda ash manufacturing process?
It is an industrial system used to produce sodium carbonate through chemical synthesis or natural ore processing.
2. What raw materials are used?
Common inputs include salt, limestone, ammonia, carbon dioxide, and trona ore.
3. Which method is most common?
The Solvay process is widely used in synthetic production systems.
4. Is natural production better than synthetic?
Natural trona-based systems often produce less waste, but both methods are widely used.
5. Why is soda ash important industrially?
It is a core chemical used in glass, chemicals, and manufacturing industries worldwide.
Conclusion
The soda ash manufacturing process is a complex yet highly efficient industrial system that transforms raw materials into one of the most widely used chemical compounds in the world. From synthetic Solvay systems to natural trona extraction, each soda ash production process plays a critical role in global supply chains.
Understanding the sodium carbonate manufacturing process highlights how chemistry, engineering, and industrial logistics work together to meet global demand. With continuous innovation and sustainable improvements, the industry is moving toward cleaner and more efficient production systems.
Companies like Basekim continue to support industrial markets by ensuring reliable supply and distribution channels for this essential material, reinforcing the global importance of this manufacturing ecosystem.